Topic 2: Atomic Structure
|
|
Whats importantModeling atom
Electronic configuration Spectral analysis SPDF block periodic table |
IF YOU DO NOT UNDERSTAND ANY TERMS! CLICK ME FOR ALL VOCAB!!!
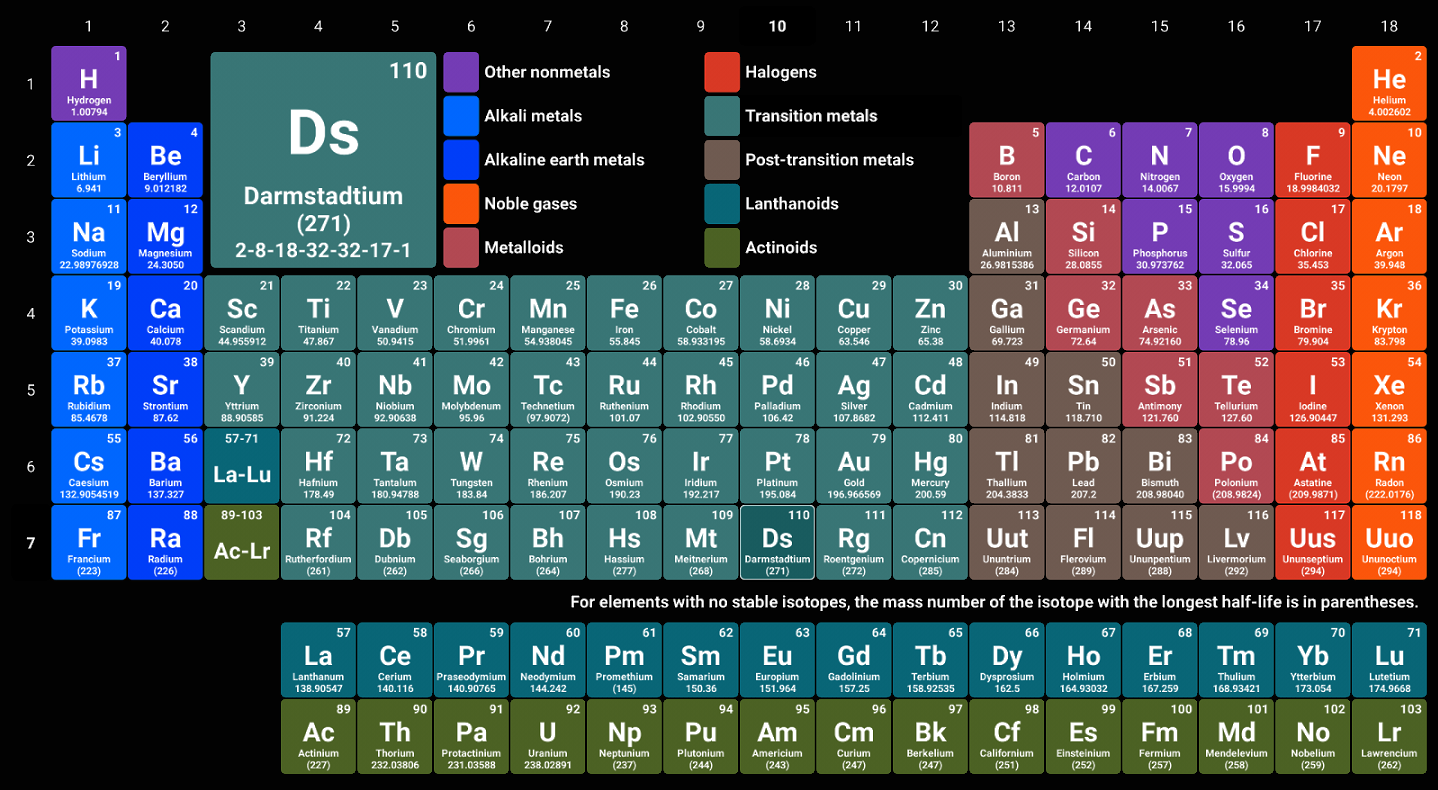
What are the periodic table blocks? |
They are groups of atoms, labeled SPFD, which share similar electronic configurations
For example, S block in orange, relates to the S orbital which holds up to two electrons. |
How does this help me? |
Once you get good at recognizing the different blocks it becomes easier to navigate the periodic table, and to determine the electronic configuration of different atoms.
|
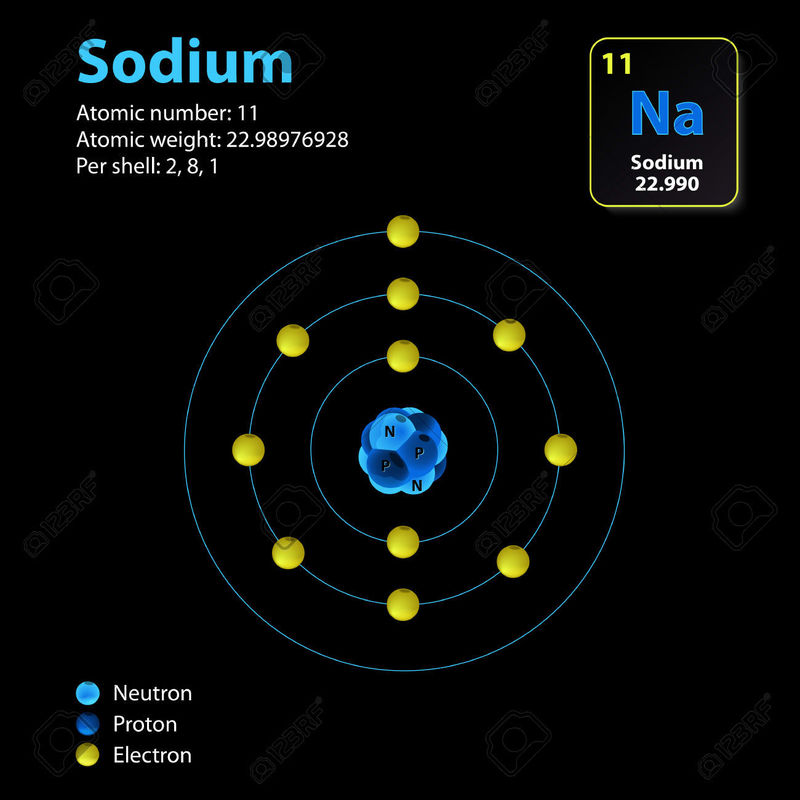
INSIDE THE ATOM
|
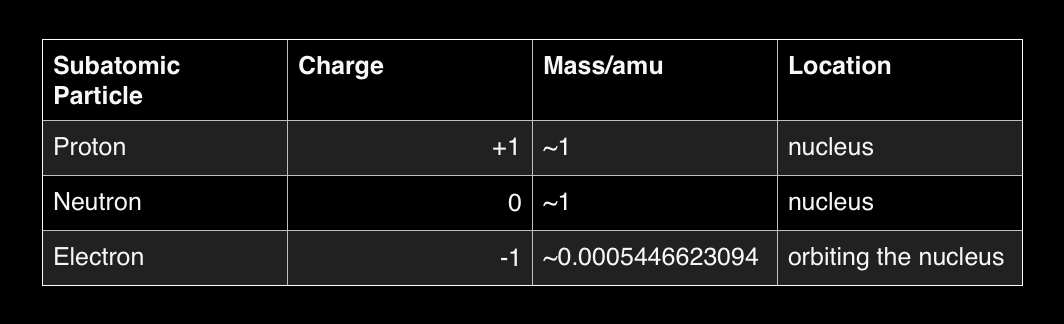
While this is a two dimensional diagram, for the purposes of this chapter it is sufficient, The atom's nucleus is made of Protons and Neutrons. It is surrounded by orbiting electrons which have almost no mass, yet hold a full negative charge equivalent to the positive charge of a proton.
Now that we know what is in an atom, how do we determine which atom is which?
The answer is..... |
Spectroscopy!
This is the study of the range of light and radiation that helps us determine the composition of everything from distant galaxies, the composition of our sun, to the gas that we are currently breathing.
What is a Mass Spectrometer?
This will explain the relationship to orbitals and wavelength of light produced! |
Essentially it is a tool that allows us to see many waveforms of light, and split them into their base colors so we can see what waveform of light is being reflected back to us.
Well, as previously said, the spectrograph shows different colors for different elements, like Hydrogen below is red, blue, violet, and blue-green. The colors represent the release of energy in the form of photons, as the electrons move in their orbitals, falling from one to the next. The electrons then begin to rise to the next orbital plane by absorbing light, and then releasing it when they fall. This video to the left should really help! |
THE BATTLE OF THE ORBITALS
Once upon a time there was a kingdom of water molecules... They were hated by many hydrophones but none more than Francium. The Francium molecules despised the water molecules for being stable and acting noble. Therefore when the Francium molecules would come across a water molecule they would fight to the death until they became unstable and would explode in flames. The francium would eat ⅔ of the water leaving only solitary hydrogen to fend for itself.... The greedy Francium now is not longer a single element, but a molecule that has the dream to combine with more, and more molecules.... Leading us to continue our story elsewhere... somewhere else on the periodic table of awesomeness.